|
Remember that all the alkali metals react vigorously with halogens to produce salts, as the halogens needs one electron to complete its octet and alkali metals can readily lose electrons to have an oxidation state of $ +1. Cesium is second from the bottom of this group, with six electron shells, so it fits all the characteristics of a reactive atom, therefore making it the most reactive element. Down to the Francium the reactivity must be slightly decreased due to the heavy nuclei.Īlkali metals (situated far away from transitional metals and noble gases) are the most reactive element group. There is no relation of radioactivity and tendency to forming compounds. This is what causes these elements to react in the same ways as the other members of the family. All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. Due to that heavy nuclei the interactions between the nucleus and the electron is more and hence it requires high energy to remove an electron from the outermost shell. Francium is most reactive element in periodic table. Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group. But it is not the case, Francium has a heavier nucleus than Cesium and it causes radioactivity. $Īs we know that reactivity increases down the group, so, you must be thinking that Francium is looking more reactive. They tend to donate their electrons in reactions and have an oxidation state of $ +1. Alkali metals are among the most reactive metals in the periodic table and this is due to their larger atomic radii and low ionization energies. 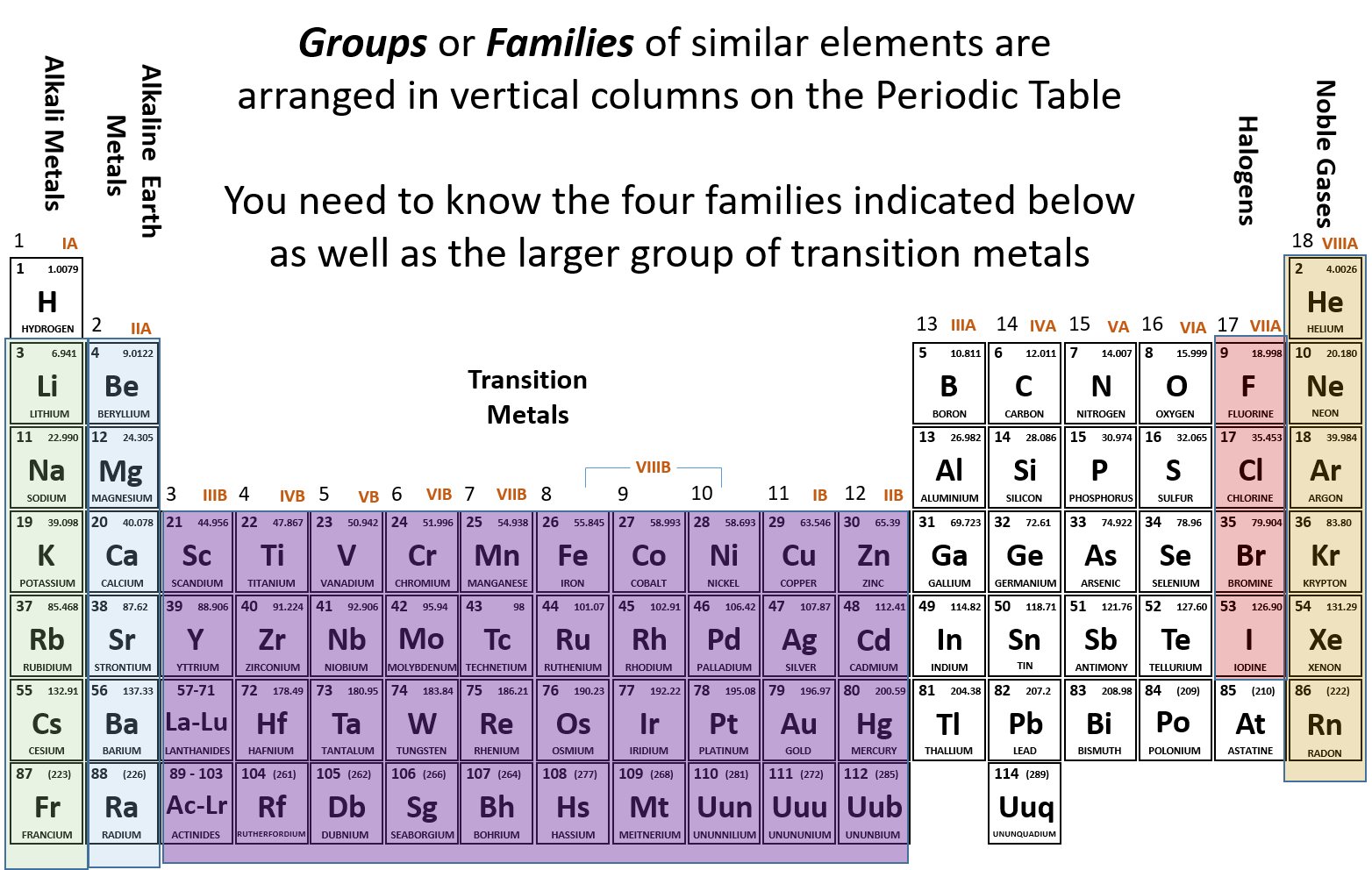 
Reactivity of elements increases down the group. They easily react with water to form an alkaline metal hydroxide solution and hydrogen. The elements are generally soft, reactive metals with low melting points. For example, Noble gases have low reactivity because they have full electron shells. Hint :We know that the number of electrons in the outermost shell of an atom and its tendency to lose electrons determines its reactivity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
